ApoB is the primary protein found on every atherogenic lipoprotein particle (LDL, VLDL, IDL, Lp(a)) — one particle, one ApoB molecule. This makes it a direct particle count, which is mechanistically more informative than LDL cholesterol concentration alone.
The 2024 National Lipid Association Expert Consensus proposed stratified ApoB targets: <90 mg/dL for intermediate-risk, <70 mg/dL for high-risk, and <60 mg/dL for very high-risk patients — thresholds consistent with epidemiological data and treatment effects observed in lipid-lowering trials. In a 2024 analysis of over 500,000 UK Biobank participants, when LDL-C and ApoB were discordant, cardiovascular risk followed ApoB — not LDL-C. Elevated ApoB was also independently associated with increased Alzheimer's disease risk.
References:
Handelsman Y, et al. Consensus Statement by the American Association of Clinical Endocrinology on the Management of Dyslipidemia. Endocr Pract. 2024.
Ibanez B, et al. Meta-analysis of LDL-C and atherosclerosis in optimal-risk individuals. J Am Coll Cardiol. 2021.

The longevity field's departure from standard LDL-C targets is partly a shift in what is measured — toward particle number (ApoB) — and partly a lowering of the absolute threshold. A key meta-analysis demonstrated that even in individuals with all cardiovascular risk factors in the optimal range, there was a significant correlation between LDL-C concentration and the presence of atherosclerosis.
Ibanez et al. (2021): In individuals with optimal values for all cardiovascular risk factors, there wa s a significant correlation between LDL-C concentration and subclinical atherosclerosis — suggesting no truly safe LDL-C level exists from a plaque standpoint.
References:
Ibanez B, et al. Subclinical atherosclerosis and LDL-C in optimal-risk individuals. J Am Coll Cardiol. 2021;77(24):3031–3041.
Ridker PM, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein (JUPITER). N Engl J Med. 2008;359(21):2195–207.

Unlike every other biomarker in this article, Lp(a) cannot be modified by lifestyle. Lp(a) levels are approximately 70–90% genetically determined and remain stable throughout life — making elevated Lp(a) a lifelong cardiovascular risk factor. The longevity field treats it as a foundational one-time test that reframes lifetime risk and justifies more aggressive management of all other modifiable factors.
Copenhagen General Population Study (Mendelian randomization, n=40,486): genetically elevated Lp(a) was associated with a 22% increased risk of myocardial infarction per doubling of Lp(a) levels.
Participant-level meta-analysis of 6 placebo-controlled statin trials: Lp(a) and LDL-C independently and additively predict ASCVD risk — meaning elevated Lp(a) confers significant residual risk even in well-controlled LDL-C patients.
The EAS 2022 consensus: the association between Lp(a) and ASCVD outcomes is continuous. The clinical cutoffs of <30 mg/dL and >50 mg/dL are pragmatic rather than biological. An Lp(a) of 100 mg/dL approximately doubles ASCVD risk, with the absolute increase substantially greater in higher-baseline-risk individuals.
Emerging therapy: Pelacarsen (antisense oligonucleotide) reduced Lp(a) by up to 80% at the highest dose in phase 2 trials, with 98% of patients achieving levels below 50 mg/dL. The phase 3 Lp(a)HORIZON cardiovascular outcomes trial is ongoing.
The 2024 NLA focused update recommends measuring Lp(a) at least once in every adult — a position now shared by the EAS, ACC/AHA, and ESC.
References:
Nordestgaard BG, et al. Lipoprotein(a) as a cardiovascular risk factor: current status. Eur Heart J. 2010;31(23):2844–53.
Tsimikas S, et al. Antisense therapy targeting apolipoprotein(a): a randomised, double-blind, placebo-controlled phase 1 study. Lancet. 2015;386(10002):1472–83.
Ridker PM, et al. Lipoprotein(a), LDL-C, and hs-CRP and 30-year cardiovascular outcomes in women. N Engl J Med. 2024.
Handelsman Y, et al. 2024 NLA Focused Update. J Clin Lipidol. 2024.

HbA1c reflects average blood sugar over the past 2–3 months. Centenarian studies consistently show HbA1c below 6% throughout life, with the lowest all-cause mortality in non-diabetic populations observed in the 5.0–5.6% range (NHANES III data). The longevity-optimized target of 5.0–5.4% reflects this evidence. Note: the lower bound of 5.0% is important — NHANES data shows a U-shaped relationship in non-diabetics, with HbA1c below5.0% associated with increased mortality, likely reflecting anemia, liver disease, or other confounding illness rather than beneficial metabolic control.
NHANES III dataset: participants without diabetes and HbA1c in the 5.0–5.6% range represented the lowest all-cause mortality reference category. Undiagnosed diabetes (HbA1c >6.5%) carried a 1.3-fold greater all-cause mortality risk.
Centenarian cohort (Catalonia): biomarkers related to glucose metabolism — including HbA1c and fasting glucose — had a significant negative impact on the probability of surviving to extreme old age when in the highest quintiles.
Important nuance: The ACCORD RCT found that intensive pharmacological lowering of HbA1c below 6.5% in established type 2 diabetics increased mortality — attributed to hypoglycemia from aggressive drug therapy. The longevity goal is naturally low HbA1c through metabolic health, not pharmacological suppression in frail or diabetic patients.
References:
Selvin E, et al. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med. 2010;362(9):800–11.
ACCORD Study Group. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med. 2008;358(24):2545–59.
Ponjoan A, et al. Centenarian biomarker profile in a population-based cohort. GeroScience. 2023.
Carson AP, Fox CS, McGuire DK, et al. Low Hemoglobin A1c and Risk of All-Cause Mortality Among US Adults Without Diabetes. Circ Cardiovasc Qual Outcomes. 2010;3(6):661-667

Standard laboratory reference ranges for fasting insulin are notably permissive. Today's lab reference ranges do not define hyperinsulinemia until fasting insulin rises above 24.9 µIU/mL — which is three to five times higher than what longevity-focused clinicians consider optimal. Elevated fasting insulin is an earlier signal of insulin resistance than HbA1c, often appearing years before glucose dysregulation becomes apparent.
Prospective meta-analysis (Zhang et al., Bioscience Reports, 2017): each unit increase in fasting insulin within the normal range was associated with a 5% increase in cardiovascular risk. Those with fasting insulin below 5 µIU/mL had significantly lower cardiovascular and all-cause mortality. Xun et al showed that each 50-pmol/L increment in fasting insulin was associated with a 25% increase in risk of hypertension and a 16% increase in risk of CHD.
References:
Zhang X, et al. Fasting insulin, insulin resistance and risk of hypertension in the general population. Bioscience Reports. 2017;37(5):BSR20160343.
Sung KCC, Seo MHH, Rhee EJJ, Wilson AM. Elevated fasting insulin predicts the future incidence of metabolic syndrome: a 5-year follow-up study. Cardiovasc Diabetol. 2011;10(1):108. doi:10.1186/1475-2840-10-108
Xun P, Wu Y, He Q, He K. Fasting insulin concentrations and incidence of hypertension, stroke, and coronary heart disease: a meta-analysis of prospective cohort studies123. The American Journal of Clinical Nutrition. 2013;98(6):1543-1554.

While the clinical threshold for prediabetes begins at 100 mg/dL, prospective cohort data show that fasting glucose in the 90–99 mg/dL range is associated with progressively increasing cardiovascular and metabolic risk compared to levels below 90 mg/dL. The longevity field treats this zone as an early warning signal rather than a reassuring normal result.
References:
Levitan EB, et al. Is nondiabetic hyperglycemia a risk factor for cardiovascular disease? Arch Intern Med. 2004;164(19):2147–55.
Rijkelijkhuizen JM, et al. Optimal cutoff points for fasting glucose and related cardiovascular risk. Diabetes Care. 2007;30(5):1167–72.
Bao C, Zhang D, Sun B, et al. Optimal Cut-Off Points of Fasting Plasma Glucose for Two-Step Strategy in Estimating Prevalence and Screening Undiagnosed Diabetes and Pre-Diabetes in Harbin, China. PLOS ONE. 2015;10(3):e0119510.
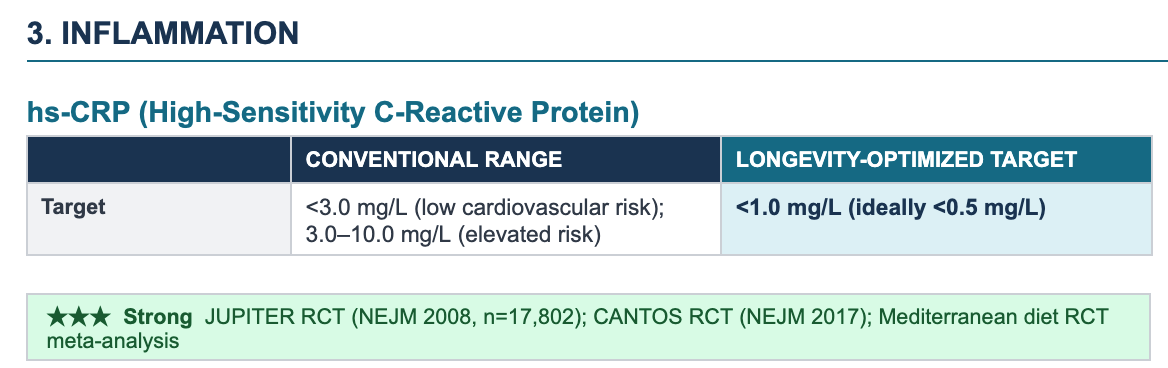
The pivotal RCT evidence comes from the landmark JUPITER trial: a randomized, placebo-controlled study of 17,802 apparently healthy men and women that established hs-CRP as an independent, actionable cardiovascular risk marker even in patients with normal LDL-C.
JUPITER within-trial dose-response analysis: participants who achieved hs-CRP below 1 mg/L on rosuvastatin saw a 79% reduction in cardiovascular events — substantially more than those who achieved LDL-C reduction alone, firmly establishing <1 mg/L as the longevity-optimized target.
Meta-analysis of Mediterranean diet RCTs: the Mediterranean diet reduced hs-CRP by 0.98 mg/L on average; a high-fiber diet reduced hs-CRP by 28% — comparable in magnitude to treatment with lovastatin 20 mg/day.
Centenarian data (PMC 2025): centenarian populations show significantly lower plasma CRP levels (0.9 ± 0.4 mg/L) compared to average-aging elderly (2.8 ± 1.6 mg/L), with differences persisting after controlling for comorbidities.
References:
Ridker PM, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein (JUPITER). N Engl J Med. 2008;359(21):2195–207.
Ridker PM, Danielson E, Fonseca FA, et al. Reduction in C-reactive protein and LDL cholesterol and cardiovascular event rates after initiation of rosuvastatin: a prospective study of the JUPITER trial. The Lancet. 2009;373(9670):1175-1182. doi:10.1016/S0140-6736(09)60447-5
Ridker PM, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease (CANTOS). N Engl J Med. 2017;377(12):1119–31.
Ridker PM, et al. C-reactive protein levels and outcomes after statin therapy. N Engl J Med. 2005;352(1):20–8.
Keshani M, Rafiee S, Heidari H, Rouhani MH, Sharma M, Bagherniya M. Mediterranean Diet Reduces Inflammation in Adults: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Nutr Rev. Published online November 10, 2025:nuaf213. doi:10.1093/nutrit/nuaf213
Adibi S. A Conceptual Digital Health Framework for Longevity Optimization: Inflammation-Centered Approach Integrating Microbiome and Lifestyle Data—A Review and Proposed Platform. Nutrients. 2026;18(2):231.

Prospective data consistently link homocysteine above 9–10 µmol/L with cardiovascular and neurodegenerative mortality. However, the evidence base for homocysteine is more complex than other biomarkers in this article.
Nygård et al. (NEJM, 1997): in patients with angiographically confirmed coronary artery disease, homocysteine levels of 9.0–14.9 µmol/L were already associated with a mortality ratio of 3.3 compared to levels below 9 µmol/L — rising to 9.9 for levels ≥20 µmol/L.
PMC meta-analysis of 12 prospective studies (n=23,623): comparing highest to lowest homocysteine categories, CHD mortality increased 66%, cardiovascular mortality 68%, and each 5 µmol/L increment was associated with a 52% increase in CHD mortality.
Critical RCT caveat: Landmark trials — HOPE-2 (2006), NORVIT (2006), and VITATOPS (2010) — successfully lowered plasma homocysteine with B-vitamin supplementation but largely failed to reduce hard cardiovascular endpoints including MI, stroke, or all-cause mortality. A subsequent meta-analysis of 19 RCTs (n=47,921) found B vitamin supplementation significantly reduced stroke risk but not CVD, MI, CHD, or all-cause mortality. This does not negate homocysteine's value as a risk biomarker, but cautions against assuming pharmacological normalization alone improves outcomes.
References:
Nygård O, et al. Plasma homocysteine levels and mortality in patients with coronary artery disease. N Engl J Med. 1997;337(4):230–6.
Lonn E, et al. Homocysteine-lowering therapy and outcomes in high-risk patients (HOPE-2). N Engl J Med. 2006;354(15):1567–77.
Clarke R, et al. Effects of lowering homocysteine levels with B vitamins on cardiovascular disease, cancer, and cause-specific mortality: meta-analysis of 8 randomized trials. Arch Intern Med. 2010;170(18):1622–31.
Homocysteine Studies Collaboration. Homocysteine and risk of ischemic heart disease and stroke: a meta-analysis. JAMA. 2002;288(16):2015-2022. doi:10.1001/jama.288.16.2015
Peng H et al. Elevated homocysteine levels and risk of cardiovascular and all-cause mortality: a meta-analysis of prospective studies. J Zhejiang Univ Sci B. 2015;16(1):78-86.

IL-6 is the most studied pro-inflammatory cytokine in the context of biological aging ("inflammaging"). Standard lab reference ranges are population-derived and do not reflect the levels associated with optimal longevity outcomes.
Centenarian cohort data (PMC 2025): centenarians demonstrate significantly reduced plasma IL-6 (1.4 ± 0.6 pg/mL) compared to average-aging elderly individuals (3.2 ± 1.8 pg/mL, p<0.001). These differences persist after controlling for comorbidities, suggesting low inflammatory status is a characteristic feature of exceptional longevity.
Tylutka et al., Frontiers in Immunology (2024, PRISMA meta-analysis): the overall IL-6 concentration was significantly higher in patients with age-related diseases compared to controls (SMD 0.16, p<0.001). IL-6 demonstrated diagnostic utility as an independent predictor of age-related morbidity and mortality in elderly adults.
Mendelian randomization (npj Genomic Medicine, 2019): IL-6 signaling pathway showed causal inverse associations with atrial fibrillation, stroke, coronary artery disease, and abdominal aortic aneurysm — and was positively associated with parental age at death (longevity proxy).
References:
Tylutka A, et al. Level of IL-6, TNF, and IL-1β and age-related diseases: a systematic review and meta-analysis. Front Immunol. 2024;15:1330386.
Collaboration (Centenarian data). A Conceptual Digital Health Framework for Longevity Optimization. PMC 2025.
Georgakis MK, et al. IL-6 signaling in cardiovascular diseases, immune-related disorders and longevity. npj Genomic Medicine. 2019.
Adibi S. A Conceptual Digital Health Framework for Longevity Optimization: Inflammation-Centered Approach Integrating Microbiome and Lifestyle Data—A Review and Proposed Platform. Nutrients. 2026;18(2):231.

IGF-1 is more nuanced than most biomarkers — higher is decidedly not better. The relationship with longevity is U-shaped: both very low and very high levels carry increased risk, with the middle range associated with lowest mortality.
Longo et al., Aging Cell (2022) — meta-analysis of 19 prospective cohort studies (n=30,876): dose-response analysis revealed a U-shaped relation between IGF-1 and all-cause mortality HR. The 120–160 ng/mL range was associated with lowest mortality. Comparing low vs. middle IGF-1 showed a significantly increased all-cause mortality (HR=1.33, 95% CI: 1.14–1.57); high vs. middle also showed elevated mortality (HR=1.23, 95% CI: 1.06–1.44).
EPIC-Heidelberg (Mukama et al., J Clin Endocrinol Metab, 2023, population-based prospective study): higher IGF-1 showed direct associations with breast cancer (HR 1.25) and prostate cancer (HR 1.31) risk. Both lowest and highest IGF-1 levels were at increased risk of deaths from cancer, CVD, and all causes — confirming the U-shaped relationship in a large European population.
The practical takeaway: aim for a physiologically mid-range IGF-1 — typically achieved through adequate but not excessive protein intake, regular exercise, and healthy sleep — not pharmacological optimization of GH/IGF-1 axis.
References:
Rahmani J, et al. Association between IGF-1 levels ranges and all-cause mortality: a meta-analysis. Aging Cell. 2022;21(2):e13540.
Mukama T, et al. IGF-1 and risk of morbidity and mortality from cancer, cardiovascular diseases, and all causes in EPIC-Heidelberg. J Clin Endocrinol Metab. 2023;108(10):e1092–e1105.

The most robust evidence comes from a 2023 Lancet Diabetes & Endocrinology IPD meta-analysis — one of the largest thyroid function studies ever conducted — which explicitly set out to define optimal TSH and FT4 ranges based on clinical outcomes rather than statistical population percentiles.
Xu et al., Lancet Diabetes & Endocrinology (2023), n=134,346, avg. follow-up 11.5 years: nonlinear associations identified for TSH, with the 60th–80th percentiles (median 1.90–2.90 mIU/L) associated with the lowest risk of CVD and mortality. FT4 showed a J-shaped association with the 20th–40th percentile (median 13.5–14.8 pmol/L) conveying the lowest risk. FT4 in the 80th–100th percentiles was associated with a 1.34-fold increase in all-cause mortality and a 1.57-fold increase in CVD mortality compared to the optimal range.
Interpretation: The longevity-optimal TSH is neither at the low end (which is associated with subclinical hyperthyroidism and elevated CVD/mortality risk) nor at the conventional upper limit of 4.0 mIU/L. The TSH range 1.9–2.9 mIU/L — which most clinicians would currently consider unremarkably normal — is where mortality risk is lowest. For older individuals especially, higher-normal TSH may be adaptive rather than pathological.
References:
Xu Y, et al. The optimal healthy ranges of thyroid function defined by the risk of cardiovascular disease and mortality: systematic review and individual participant data meta-analysis. Lancet Diabetes Endocrinol. 2023;11(10):743–754. PMID: 37696273.
Razvi S, et al. The beneficial effect of L-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism. J Clin Endocrinol Metab. 2007;92(5):1715–23.

Standard labs flag 'low' testosterone only at the population floor — a statistical, not a health-optimized threshold. Longevity medicine is less interested in avoiding 'low' and more focused on maintaining levels associated with metabolic health, muscle mass preservation, insulin sensitivity, and cognitive protection — typically the upper-mid range for age.
The Testosterone Trials (TTrials, NEJM 2016): in men 65+ with low testosterone (<275 ng/dL), testosterone treatment improved sexual function, physical function, and quality of life, with additional signals for bone density and anemia. Metabolic benefits were observed at levels in the mid-normal range.
References:
Snyder PJ, et al. Effects of testosterone treatment in older men (TTrials). N Engl J Med. 2016;374(7):611–24.
Araujo AB, Dixon JM, Suarez EA, Murad MH, Guey LT, Wittert GA. Clinical review: Endogenous testosterone and mortality in men: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2011;96(10):3007-3019. doi:10.1210/jc.2011-1137

Conventional medicine only acts on eGFR below 60 mL/min/1.73m². The longevity field recognizes that even subclinical decline within the 60–89 range significantly elevates cardiovascular and all-cause mortality risk, making >90 an active target rather than an irrelevant benchmark.
CKD Prognosis Consortium (Lancet, 2010, n=>1.1 million): there was a continuous, independent, and graded association of both reduced eGFR and increased albuminuria with all-cause mortality, cardiovascular mortality, and kidney failure — with significant excess risk beginning below eGFR 75 mL/min/1.73m², not only at the CKD threshold of 60.
References:
Matsushita K, et al. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet. 2010;375(9731):2073–81.

Clinical trial data on vitamin D have historically been mixed, but evidence for the 40–60 ng/mL target has strengthened considerably with recent RCTs.
TARGET-D RCT (2024): in high-risk patients, vitamin D supplementation titrated to levels above 40 ng/mL significantly reduced the risk of recurrent heart attacks — one of the first RCTs to demonstrate a cardiovascular benefit tied specifically to achieving a target level above 40 ng/mL.
VITAL RCT (NEJM, 2019, n=25,871): while the primary endpoints were not significantly reduced, pre-specified analyses showed significant reductions in cancer mortality in participants with low BMI — with post-hoc analyses consistently suggesting greater benefit when 25-OH-D levels were maintained above 40 ng/mL.
References:
Manson JE, et al. Vitamin D supplements and prevention of cancer and cardiovascular disease (VITAL). N Engl J Med. 2019;380(1):33–44.
Scragg R, et al. Effect of monthly high-dose vitamin D supplementation on cardiovascular disease in the ViDA trial. JAMA Cardiol. 2017;2(6):608–16.
TARGET-D Trial. Titrated vitamin D supplementation and cardiovascular outcomes. 2024.

Longevity medicine watches both ends of the ferritin range. Chronically elevated ferritin — even within the conventional 'normal' upper limit — is associated with oxidative stress, iron overload, non-alcoholic fatty liver disease, and cardiovascular risk.
UK Biobank and prospective cohort data: serum ferritin in the upper normal range (>200 ng/mL in men) was associated with increased risk of liver disease, metabolic syndrome, cardiovascular events, and all-cause mortality — independent of CRP — suggesting true iron overload rather than inflammatory ferritin elevation as a contributor.
References:
Ellervik C, Mandrup-Poulsen T, Andersen HU, et al. Elevated Transferrin Saturation and Risk of Diabetes: Three population-based studies. Diabetes Care. 2011;34(10):2256-2258. doi:10.2337/dc11-0416
Ellervik C, Marott JL, Tybjærg-Hansen A, Schnohr P, Nordestgaard BG. Total and cause-specific mortality by moderately and markedly increased ferritin concentrations: general population study and metaanalysis. Clin Chem. 2014;60(11):1419-1428. doi:10.1373/clinchem.2014.229013
Mainous AG 3rd, et al. Elevated serum ferritin is associated with increased risk of type 2 diabetes. J Am Board Fam Med. 2005;18(3):174–81.
Kadoglou NPE, Biddulph JP, Rafnsson SB, Trivella M, Nihoyannopoulos P, Demakakos P. The association of ferritin with cardiovascular and all-cause mortality in community-dwellers: The English longitudinal study of ageing. PLOS ONE. 2017;12(6):e0178994. doi:10.1371/journal.pone.0178994
† Homocysteine observational signal is strong; RCT intervention evidence is weak. See Section 3 for nuance.
IMPORTANT CLINICAL CAVEATS
Several of these markers have J-shaped or U-shaped relationships with mortality (HbA1c, uric acid, IGF-1, TSH) — meaning both very high and very low values carry risk. The goal is physiological optimization, not maximizing or minimizing a number in isolation.
The longevity field increasingly distinguishes between a biomarker's epidemiological association with longevity and the RCT-supported benefit of pharmacologically altering that biomarker. For some markers (homocysteine, HbA1c in established diabetics), these diverge significantly. Where possible, optimal ranges should be pursued through lifestyle modification — diet, exercise, sleep, and metabolic health — rather than drug or supplement therapy alone.
This document reflects the evidence base as of 2024–2025. Reference ranges and therapeutic thresholds are actively evolving fields; clinicians should apply individual patient context to all targets presented here.